New Ketoproof System Flags Medications Safe for Ketogenic Therapy
Ketoproof turns a long-known keto trap into a usable screening system. It helps flag meds that quietly carry carbs and can knock patients out of ketosis.
Ketoproof tackles one of the most frustrating keto therapy failures: the medicine cabinet. A new Springer paper by Sanaa Shaaban, Mohammed Al-Beltagi, Omnia El Rashidy, May Nassar, Yasmin El Gendy, Natasha E Schoeler, Victoria J Whiteley, and Elles van der Louw turns hidden carbohydrate exposure from a vague warning into a structured medication screen.
Why this matters in real life
If you live with medical keto, you already know the drill: weigh the food, count the grams, keep the ratio tight, then hope nothing slips through the cracks. The problem is that a prescription can do what a plate of pasta never would, and do it quietly. Liquid medicines, chewables, syrups, flavorings, and other excipients can carry enough carbohydrate to undermine ketosis, especially in children with refractory epilepsy.
That is why Ketoproof lands as more than a database project. It treats ketogenic therapy as a whole-system treatment, where pharmacy, nutrition, and neurology have to stay aligned. For families who have spent months protecting a seizure-control ratio, that is the difference between a therapy that holds and one that gets derailed by a formulation detail.
What Ketoproof classifies
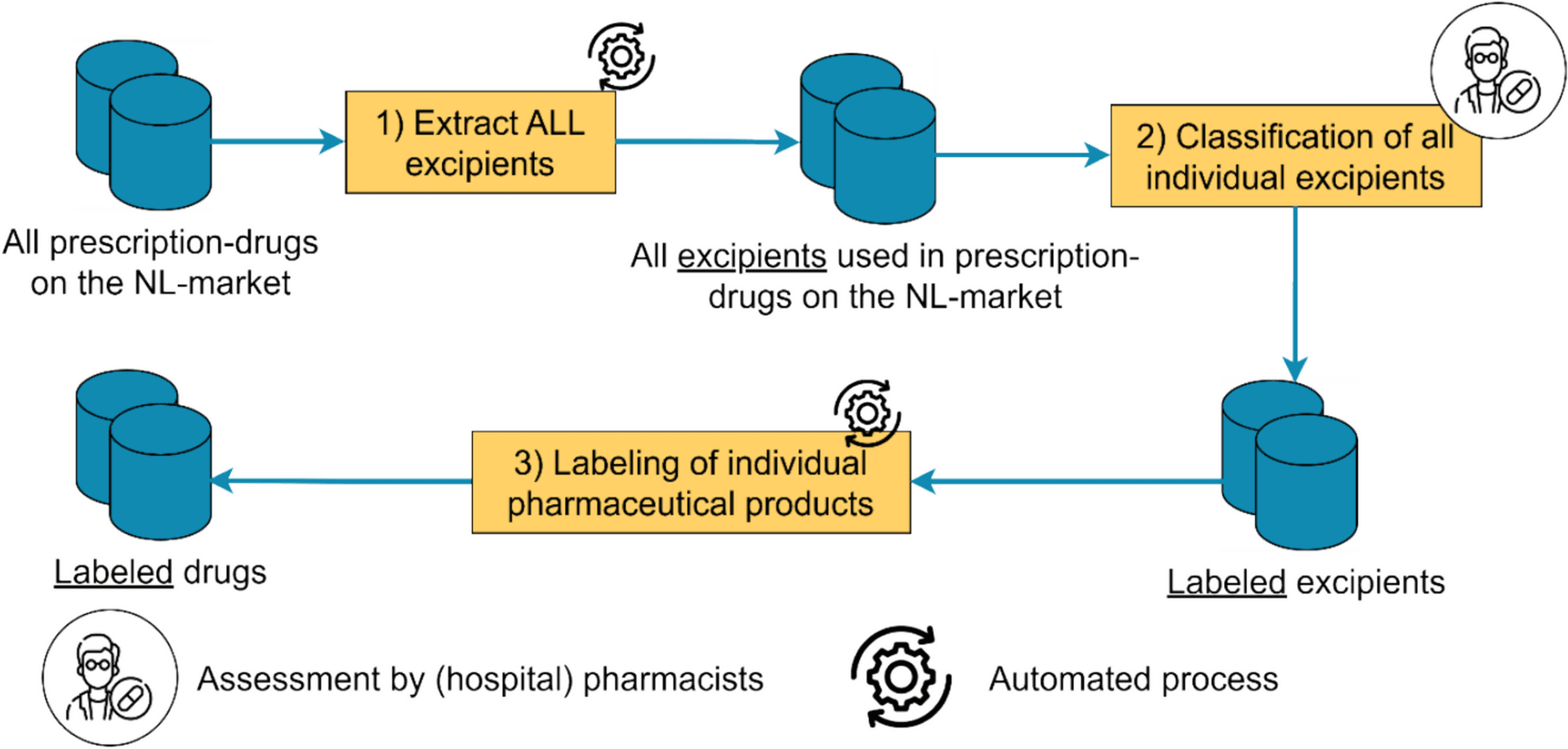
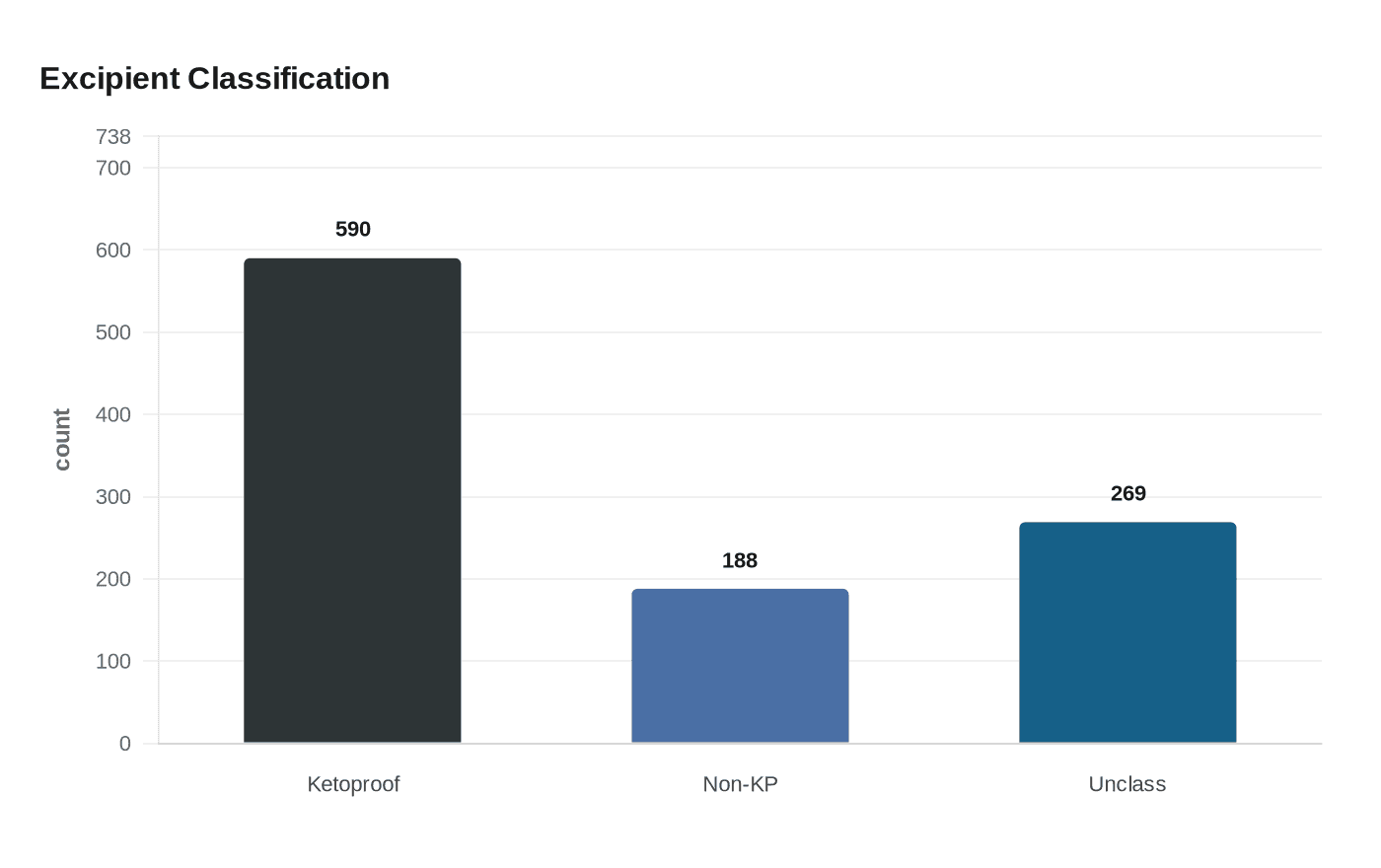
The paper introduces a reproducible framework for labeling pharmaceutical excipients used in medicines marketed in the Netherlands. The research team examined 1,047 excipients in total and successfully classified 778 of them. Of those, 590 were labeled Ketoproof and 188 were labeled non-Ketoproof, while 269 remained unclassified.
That matters because excipient review has long been the annoying hidden step in keto care. A medicine may be active-drug safe and still be wrong for keto because of syrup bases, sweeteners, or other carbohydrate-containing ingredients. Ketoproof does the unglamorous work of sorting those ingredients into a system clinicians can use consistently instead of relying on memory, brand familiarity, or a rushed label check.
When the team applied those labels to actual products, they analyzed 28,721 pharmaceutical products and found that 41 percent, or 11,781 products, were Ketoproof. That is a useful number for day-to-day care because it shows a large share of products can fit ketogenic therapy, but not by assumption. You still need to verify the exact formulation, not just the active ingredient name.
Why the Dutch medicines data source matters
The product work was built on the Netherlands Medicines Information Bank, which is managed by the Netherlands Medicines Authority, or MEB. The database is updated weekly, and its site listed the last update as April 29, 2026. That gives Ketoproof a strong practical base, because it rests on an official national medicines source rather than a grab bag of product lists.

That kind of structure is exactly what keto care has been missing. Excipients are notoriously hard to compare across manufacturers and countries, and the same active drug can come in a version that is fine for ketosis and another that is not. A framework tied to a maintained national medicines database is a lot more useful in a clinic than a spreadsheet someone built once and forgot.
How KetoMed changes the workflow
The practical output of the study is KetoMed, a web-based tool designed to help healthcare professionals review medications for people on ketogenic diet therapy. The point is not just to identify a few problematic products. It is to make medication review faster, safer, and more consistent every time a prescription changes, a new brand appears, or a family gets discharged with a stack of new orders.
For patients, caregivers, and pharmacists, that changes the rhythm of the day-to-day conversation. Instead of asking only, “Is this drug on the formulary?”, the real question becomes, “Is this formulation keto-safe, and if not, what is the lowest-risk alternative?”
A practical workflow looks like this:
1. Check the active drug and the exact dosage form.
2. Look for hidden carbohydrate sources in liquid bases, chewables, flavorings, and premixes.
3. Confirm whether the product has a Ketoproof label or needs a manual review.
4. If the medicine is not suitable, ask whether a tablet, capsule, different manufacturer, or different route avoids the carb load.
That is the sort of routine that prevents a last-minute pharmacy scramble when a child suddenly gets a new antiseizure medication or an inpatient order is rewritten at discharge.
The questions to ask before assuming a prescription is keto-compatible
This is the part readers can use immediately. A prescription is not keto-compatible just because it looks ordinary on the label. Before you take it at face value, ask:
- What is the exact formulation, not just the drug name?
- Does it contain syrups, sugar alcohols, sweeteners, flavorings, or carbohydrate-based diluents?
- Is there a tablet, capsule, or alternative brand with the same active ingredient and fewer excipients?
- If it is an IV medication, was it premixed in a carbohydrate-containing solution, or can it be given in normal saline?
- Has the product been checked against a ketogenic medication review tool, not just a standard drug reference?
Those are not theoretical questions. A 2025 study in the American Journal of Health-System Pharmacy found that hospitalized patients on ketogenic diets are at increased risk for inappropriate carbohydrate-containing medications, and it flagged IV premix products diluted in carbohydrate-containing solutions when no alternative file existed for saline dilution. That is exactly the kind of hidden hazard Ketoproof is meant to reduce.
Why pediatrics keeps driving this issue
The ketogenic diet has been used to treat children with epilepsy since the 1920s, and Johns Hopkins Medicine says its pediatric program has treated more than 1,500 children with this approach. That long history explains why this issue keeps resurfacing in epilepsy centers: the diet is old, but the pharmacy problem never stopped being modern.
Children are the hardest patients to “just switch” when a liquid medicine is a problem. A 2019 proof-of-principle study noted that liquid medications can be a source of hidden carbohydrates, and it also pointed out that moving infants and young children from liquids to tablets is not always feasible. That same study even explored whether increasing the ketogenic ratio could offset carbohydrates in liquid antiseizure drugs, which tells you how serious the problem can get when there is no easy formulation swap.
How this fits into current guidance
Ketoproof does not arrive in a vacuum. The NHS Specialist Pharmacy Service published guidance in August 2024 and updated it on November 18, 2025, on how to assess whether medicines are suitable for people on a medical ketogenic diet. Matthews Friends also maintained a carbohydrate-content-of-medicines resource updated in May 2025, showing that patient-support groups and pharmacy services have been pushing in the same direction for years.
What Ketoproof adds is standardization. Instead of scattered advice, it offers a transparent classification system that could support international consistency and better regulatory transparency around excipient content. That is especially useful in a condition where the smallest formulation change can affect seizure control.
The blunt takeaway is simple: keto therapy is not just a nutrition plan. It is medication-sensitive, formulation-sensitive, and often hospital-sensitive. Ketoproof gives clinicians a better screen, and it gives families a better shot at catching the prescription that would otherwise spoil the ratio.
Know something we missed? Have a correction or additional information?
Submit a Tip