Glucose controls brain cell development, keto rescue reveals myelin pathway
A mouse study links glucose to OPC growth and myelin timing, then shows ketones can rescue a broken pathway. For keto readers, the lesson is stage-specific biology, not a blanket rule.
Glucose is doing more than feeding the brain
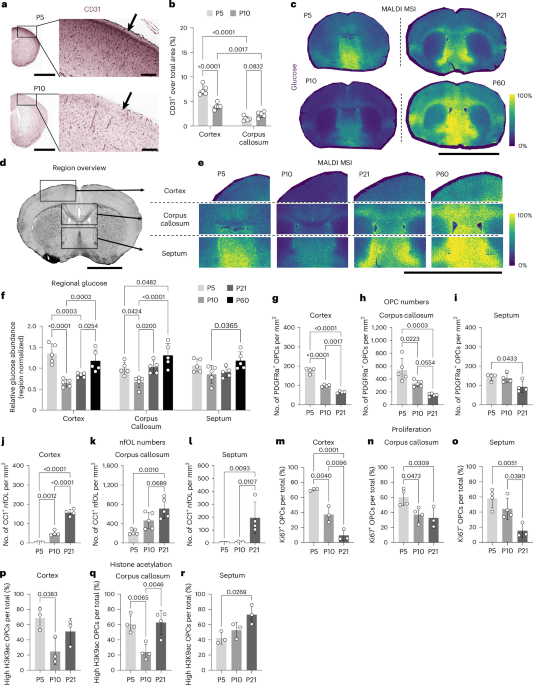
The new Nature Neuroscience paper lands right in the middle of a debate keto readers know well: is glucose just the thing you restrict, or is it also part of the signaling machinery that keeps biology on schedule? In this mouse study, glucose availability was tied directly to oligodendrocyte precursor cell, or OPC, behavior, and the effect changed by brain region and developmental timing. That matters because myelin does not appear all at once, and the paper argues that regional glucose patterns help explain why some white-matter tracts mature earlier than others.
The author list includes Sami Sauma, Stephanie Stransky, Ipek Selcen, Simone Sidoli, Rinat Abzalimov, Ye He, and Patrizia Casaccia. Their central finding is simple to state and surprisingly rich in consequences: temporal and brain-regional glucose fluctuations, along with changes in vascularization, modulate OPC population dynamics. Regions with higher glucose showed more OPC proliferation and higher histone acetylation, linking a metabolic input to an epigenetic readout that helps drive cell expansion.
Why ACLY sits at the center of the story
The key enzyme here is ACLY, which converts glucose-derived citrate into acetyl-CoA. In the study, ACLY mediated the link between higher glucose and greater OPC proliferation, making glucose act less like a passive fuel and more like a developmental cue. Lead author Sami Sauma described glucose as "not just fuel for the brain" but also a signal for cells to divide, which is the right lens for understanding why this paper complicates simple glucose-versus-ketones thinking.
When the team conditionally deleted Acly in OPCs, the mice developed a transient hypomyelination phenotype because the OPCs could not expand normally. That is the part keto readers will zero in on, because it shows the glucose-to-acetyl-CoA route is not optional during this stage of brain development. The cells still had the ability to differentiate into oligodendrocytes, but they could not build the precursor pool properly, which slowed the whole process.
The rescue experiment is the keto headline, but not the whole story
This is where the ketogenic-diet angle enters, and it is the reason the study is so interesting for the keto world. When mice lacking Acly in OPCs were placed on a ketogenic diet, blood ketone levels rose and the myelin deficits improved. That does not mean keto magically fixes every myelin problem, but it does show that ketones can partially substitute for a broken glucose-to-acetyl-CoA pathway in a developmental brain context.

The deeper biological point is stage specificity. The paper’s abstract says OPCs rely on ACLY-dependent nuclear acetyl-CoA from glucose-derived citrate, while oligodendrocytes rely on extranuclear acetyl-CoA from other sources for myelin formation. Patrizia Casaccia put it plainly: the same cell lineage interprets different metabolic signals at distinct developmental stages. In other words, the cell’s fuel preferences shift as it matures, and the developmental task changes with them.
What the cell is doing is not just metabolism, it is timing
The CUNY Graduate Center and the Advanced Science Research Center said the work may help explain why myelin forms at different times in different brain regions. That detail is more than academic. Myelination begins before birth and continues into adulthood, so a mechanism that links local glucose levels to OPC proliferation gives researchers a way to think about why some circuits get wrapped earlier than others.
The team mapped glucose levels across developing mouse brains using MALDI imaging at the CUNY ASRC MALDI Imaging Core Facility. That map, combined with the histone acetylation data, suggests glucose is part of a local developmental instruction set, not merely an energy meter. The paper’s data package backs that up with bulk RNA-seq accession GSE278709, single-cell RNA-seq accession GSE281944, and histone mass spectrometry project PXD069000.
Why oligodendrocytes still formed even after Acly loss
One of the most important nuances for keto readers is that the story is not a clean switch from glucose dependence to ketone dependence. Even after Acly deletion in OPCs, oligodendrocyte differentiation still proceeded because the cells compensated by upregulating other enzymes that generate extranuclear acetyl-CoA from alternative metabolic substrates. That means the lineage retained metabolic flexibility, even when one input was gone.
This is where the paper earns its place in the broader keto conversation. Carbohydrate availability is not just fuel, and ketones are not simply a replacement fuel. The findings suggest that different developmental stages use different metabolic inputs for different jobs, with glucose helping expand the precursor pool and alternative substrates helping later-stage myelin formation continue.

How this fits with earlier work on OPC metabolism
The 2026 study does not stand alone. A 2025 Nature paper reported that blocking low-glucose-induced AMPK activation in OPCs was important for myelination and remyelination, which points to another glucose-sensing pathway shaping white-matter biology. A 2023 mouse study found that a ketogenic diet promoted OPC proliferation in the hypothalamic median eminence by activating fatty acid oxidation, showing that ketone-rich states can support OPC behavior in at least some regions.
Taken together, these studies strengthen the idea that OPCs are metabolically plastic. They respond to glucose scarcity in one context, ketone availability in another, and regional cues all along the way. For adult keto dieters, the practical takeaway is not that more ketones automatically means more myelin, but that the nervous system uses carbohydrates and ketones at different moments for different developmental tasks.
The real keto lesson
If you strip away the headline and look at the biology, this study argues for a more sophisticated view of brain fuel. Glucose can act as a developmental signal, ACLY can translate that signal into acetyl-CoA, and ketones can rescue some of the downstream consequences when that pathway breaks. That is a much more interesting story than "glucose bad, ketones good."
For the keto community, the value here is conceptual as much as practical. The paper supports a view of metabolic flexibility in brain cells, especially in myelin-forming lineages, and it offers a mechanistic rationale for future work in preterm brain injury, white-matter disorders, and other conditions where myelin development is disrupted.
Know something we missed? Have a correction or additional information?
Submit a Tip

